
SL Paper 2
Ethane-1,2-diol, HOCH2CH2OH, has a wide variety of uses including the removal of ice from aircraft and heat transfer in a solar cell.
Ethane-1,2-diol can be formed according to the following reaction.
2CO (g) + 3H2 (g) HOCH2CH2OH (g)
(i) Deduce the equilibrium constant expression, Kc, for this reaction.
(ii) State how increasing the pressure of the reaction mixture at constant temperature will affect the position of equilibrium and the value of Kc.
Position of equilibrium:
Kc:
(iii) Calculate the enthalpy change, ΔHθ, in kJ, for this reaction using section 11 of the data booklet. The bond enthalpy of the carbon–oxygen bond in CO (g) is 1077kJmol-1.
(iv) The enthalpy change, ΔHθ, for the following similar reaction is –233.8 kJ.
2CO(g) + 3H2(g) HOCH2CH2OH (l)
Deduce why this value differs from your answer to (a)(iii).
Determine the average oxidation state of carbon in ethene and in ethane-1,2-diol.
Ethene:
Ethane-1,2-diol:
Explain why the boiling point of ethane-1,2-diol is significantly greater than that of ethene.
Ethane-1,2-diol can be oxidized first to ethanedioic acid, (COOH)2, and then to carbon dioxide and water. Suggest the reagents to oxidize ethane-1,2-diol.
Markscheme
(i)
(ii)
Position of equilibrium: moves to right OR favours product
Kc: no change OR is a constant at constant temperature
(iii)
Bonds broken: 2C≡O + 3(H-H) / 2(1077kJmol-1) + 3(436kJmol-1) / 3462 «kJ»
Bonds formed: 2(C-O) + 2(O-H) + 4(C-H) + (C-C) / 2(358kJmol-1) + 2(463kJmol-1) + 4(414kJmol-1) + 346kJmol-1 / 3644 «kJ»
«Enthalpy change = bonds broken - bonds formed = 3462 kJ - 3644 kJ =» -182 «kJ»
Award [3] for correct final answer.
Award [2 max] for «+»182 «kJ».
(iv)
in (a)(iii) gas is formed and in (a)(iv) liquid is formed
OR
products are in different states
OR
conversion of gas to liquid is exothermic
OR
conversion of liquid to gas is endothermic
OR
enthalpy of vapourisation needs to be taken into account
Accept product is «now» a liquid.
Accept answers referring to bond enthalpies being means/averages.
Ethene: –2
Ethane-1,2-diol: –1
Do not accept 2–, 1– respectively.
ethane-1,2-diol can hydrogen bond to other molecules «and ethene cannot»
OR
ethane-1,2-diol has «significantly» greater van der Waals forces
Accept converse arguments.
Award [0] if answer implies covalent bonds are broken
hydrogen bonding is «significantly» stronger than other intermolecular forces
acidified «potassium» dichromate«(VI)»/H+ AND K2Cr2O7/H+ AND Cr2O72-
OR
«acidified potassium» manganate(VII)/ «H+» KMnO4 /«H+» MnO4-
Accept Accept H2SO4 or H3PO4 for H+.
Accept “permanganate” for “manganate(VII)”.
Examiners report
Many reactions are in a state of equilibrium.
The equations for two acid-base reactions are given below.
HCO3– (aq) + H2O (l) H2CO3 (aq) + OH– (aq)
HCO3– (aq) + H2O (l) CO32– (aq) + H3O+ (aq)
The following reaction was allowed to reach equilibrium at 761 K.
H2 (g) + I2 (g) 2HI (g) ΔHθ < 0
Outline the effect, if any, of each of the following changes on the position of equilibrium, giving a reason in each case.
Identify two different amphiprotic species in the above reactions.
State what is meant by the term conjugate base.
State the conjugate base of the hydroxide ion, OH–.
A student working in the laboratory classified HNO3, H2SO4, H3PO4 and HClO4 as acids based on their pH. He hypothesized that “all acids contain oxygen and hydrogen”.
Evaluate his hypothesis.
Markscheme
Award [1 max] if both effects are correct.
Reason for increasing volume:
Accept “concentration of all reagents reduced by an equal amount so cancels out in Kc expression”.
Accept “affects both forward and backward rates equally”.
HCO3– AND H2O
species that has one less proton/H+ ion «than its conjugate acid»
OR
species that forms its conjugate acid by accepting a proton
OR
species that is formed when an acid donates a proton
Do not accept “differs by one proton/H+ from conjugate acid”.
oxide ion/O2–
insufficient data to make generalization
OR
need to consider a «much» larger number of acids
OR
hypothesis will continue to be tested with new acids to see if it can stand the test of time
«hypothesis is false as» other acids/HCl/HBr/HCN/transition metal ion/BF3 do not contain oxygen
OR
other acids/HCl/HBr/HCN/transition metal ion/BF3 falsify hypothesis
correct inductive reasoning «based on limited sample»
«hypothesis not valid as» it contradicts current/accepted theories/Brønsted-Lowry/Lewis theory
[Max 2 Marks]
Examiners report
White phosphorus is an allotrope of phosphorus and exists as P4.
An equilibrium exists between PCl3 and PCl5.
PCl3 (g) + Cl2 (g) PCl5 (g)
Sketch the Lewis (electron dot) structure of the P4 molecule, containing only single bonds.
Write an equation for the reaction of white phosphorus (P4) with chlorine gas to form phosphorus trichloride (PCl3).
Deduce the electron domain and molecular geometry using VSEPR theory, and estimate the Cl–P–Cl bond angle in PCl3.
Explain the polarity of PCl3.
Calculate the standard enthalpy change (ΔH⦵) for the forward reaction in kJ mol−1.
ΔH⦵f PCl3 (g) = −306.4 kJ mol−1
ΔH⦵f PCl5 (g) = −398.9 kJ mol−1
State the equilibrium constant expression, Kc, for this reaction.
State, with a reason, the effect of an increase in temperature on the position of this equilibrium.
Markscheme
Accept any diagram with each P joined to the other three.
Accept any combination of dots, crosses and lines.
P4 (s) + 6Cl2 (g) → 4PCl3 (l) ✔
Electron domain geometry: tetrahedral ✔
Molecular geometry: trigonal pyramidal ✔
Bond angle: 100«°» ✔
Accept any value or range within the range 91−108«°» for M3.
polar AND unsymmetrical distribution of charge
OR
polar AND dipoles do not cancel
OR
«polar as» dipoles «add to» give a «partial» positive «charge» at P and a «partial» negative «charge» at the opposite/Cl side of the molecule ✔
Accept “polar AND unsymmetrical molecule”.
«−398.9 kJ mol−1 − (−306.4 kJ mol−1) =» −92.5 «kJ mol−1» ✔
«Kc =» ✔
«shifts» left/towards reactants AND «forward reaction is» exothermic/ΔH is negative ✔
Examiners report
A mixture of 1.00 mol SO2(g), 2.00 mol O2(g) and 1.00 mol SO3(g) is placed in a 1.00 dm3 container and allowed to reach equilibrium.
2SO2(g) + O2(g) 2SO3(g)
Distinguish between the terms reaction quotient, Q, and equilibrium constant, Kc.
The equilibrium constant, Kc, is 0.282 at temperature T.
Deduce, showing your work, the direction of the initial reaction.
Markscheme
Q: non-equilibrium concentrations AND Kc: equilibrium concentrations
OR
Q: «measured» at any time AND Kc: «measured» at equilibrium
[1 mark]
«Q = » = 0.500
reverse reaction favoured/reaction proceeds to the left AND Q > Kc/0.500 > 0.282
Do not award M2 without M1.
[2 marks]
Examiners report
PCl5(g) and Cl2(g) were placed in a sealed flask and allowed to reach equilibrium at 200 °C. The enthalpy change, ΔH, for the decomposition of PCl5(g) is positive.
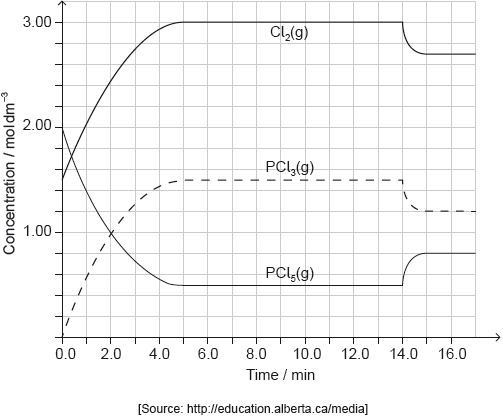
Deduce the equilibrium constant expression, Kc, for the decomposition of PCl5(g).
Deduce, giving a reason, the factor responsible for establishing the new equilibrium after 14 minutes.
Deduce the Lewis (electron dot) structure and molecular geometry of PCl3.
Markscheme
«Kc» =
[1 mark]
decrease in temperature
endothermic «reaction» AND «equilibrium» shifts to the left/reactants
OR
endothermic «reaction» AND Kc decreases
OR
endothermic «reaction» AND concentration of PCl5 increased/concentration of PCl3 and Cl2 decreased
OR
«equilibrium» shifts in exothermic direction
Do not accept “temperature change”.
Accept “ΔH positive” in place of “endothermic”.
Accept “products” instead of “PCl3 and Cl2”.
[2 marks]
Lewis structure:
Molecular geometry:
trigonal/triangular pyramidal
Penalize missing lone pairs once only between this question and 4(b).
Accept any combination of lines, dots or crosses to represent electrons.
Do not apply ECF.
[2 marks]
Examiners report
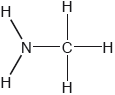
Two hydrides of nitrogen are ammonia and hydrazine, N2H4. One derivative of ammonia is methanamine whose molecular structure is shown below.
Hydrazine is used to remove oxygen from water used to generate steam or hot water.
N2H4(aq) + O2(aq) → N2(g) + 2H2O(l)
The concentration of dissolved oxygen in a sample of water is 8.0 × 10−3 gdm−3.
Estimate the H−N−H bond angle in methanamine using VSEPR theory.
Ammonia reacts reversibly with water.
NH3(g) + H2O(l) NH4+(aq) + OH−(aq)
Explain the effect of adding H+(aq) ions on the position of the equilibrium.
Hydrazine reacts with water in a similar way to ammonia. Deduce an equation for the reaction of hydrazine with water.
Outline, using an ionic equation, what is observed when magnesium powder is added to a solution of ammonium chloride.
Hydrazine has been used as a rocket fuel. The propulsion reaction occurs in several stages but the overall reaction is:
N2H4(l) → N2(g) + 2H2(g)
Suggest why this fuel is suitable for use at high altitudes.
Determine the enthalpy change of reaction, ΔH, in kJ, when 1.00 mol of gaseous hydrazine decomposes to its elements. Use bond enthalpy values in section 11 of the data booklet.
N2H4(g) → N2(g) + 2H2(g)
The standard enthalpy of formation of N2H4(l) is +50.6 kJmol−1. Calculate the enthalpy of vaporization, ΔHvap, of hydrazine in kJmol−1.
N2H4(l) → N2H4(g)
(If you did not get an answer to (f), use −85 kJ but this is not the correct answer.)
Calculate, showing your working, the mass of hydrazine needed to remove all the dissolved oxygen from 1000 dm3 of the sample.
Calculate the volume, in dm3, of nitrogen formed under SATP conditions. (The volume of 1 mol of gas = 24.8 dm3 at SATP.)
Markscheme
107°
Accept 100° to < 109.5°.
Literature value = 105.8°
[1 mark]
removes/reacts with OH−
moves to the right/products «to replace OH− ions»
Accept ionic equation for M1.
[2 marks]
N2H4(aq) + H2O(l) N2H5+(aq) + OH–(aq)
Accept N2H4(aq) + 2H2O(l) N2H62+(aq) + 2OH–(aq).
Equilibrium sign must be present.
[1 mark]
bubbles
OR
gas
OR
magnesium disappears
2NH4+(aq) + Mg(s) → Mg2+(aq) + 2NH3(aq) + H2(g)
Do not accept “hydrogen” without reference to observed changes.
Accept "smell of ammonia".
Accept 2H+(aq) + Mg(s) → Mg2+(aq) + H2(g)
Equation must be ionic.
[2 mark]
no oxygen required
[1 mark]
bonds broken:
E(N–N) + 4E(N–H)
OR
158 «kJmol–1» + 4 x 391 «kJmol–1» / 1722 «kJ»
bonds formed:
E(N≡N) + 2E(H–H)
OR
945 «kJmol–1» + 2 x 436 «kJmol–1» / 1817 «kJ»
«ΔH = bonds broken – bonds formed = 1722 – 1817 =» –95 «kJ»
Award [3] for correct final answer.
Award [2 max] for +95 «kJ».
[3 marks]
OR
ΔHvap= −50.6 kJmol−1 − (−95 kJmol−1)
«ΔHvap =» +44 «kJmol−1»
Award [2] for correct final answer.
Award [1 max] for −44 «kJmol−1».
Award [2] for:
ΔHvap − = 50.6 kJmol−1 − (−85 kJmol−1) + = 34 «kJmol−1».
Award [1 max] for −34 «kJmol−1».
[2 marks]
total mass of oxygen «= 8.0 x 10–3 gdm–3 x 1000 dm3» = 8.0 «g»
n(O2) «» 0.25 «mol»
OR
n(N2H4) = n(O2)
«mass of hydrazine = 0.25 mol x 32.06 gmol–1 =» 8.0 «g»
Award [3] for correct final answer.
[3 marks]
«n(N2H4) = n(O2) » 0.25 «mol»
«volume of nitrogen = 0.25 mol x 24.8 dm3mol–1» = 6.2 «dm3»
Award [1] for correct final answer.
[1 mark]
Examiners report
Urea, (H2N)2CO, is excreted by mammals and can be used as a fertilizer.
Calculate the percentage by mass of nitrogen in urea to two decimal places using section 6 of the data booklet.
Suggest how the percentage of nitrogen affects the cost of transport of fertilizers giving a reason.
The structural formula of urea is shown.
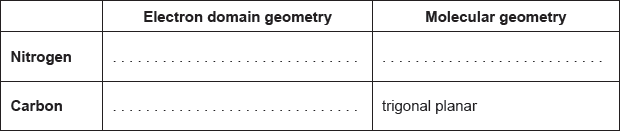
Predict the electron domain and molecular geometries at the nitrogen and carbon atoms, applying the VSEPR theory.
Urea can be made by reacting potassium cyanate, KNCO, with ammonium chloride, NH4Cl.
KNCO(aq) + NH4Cl(aq) → (H2N)2CO(aq) + KCl(aq)
Determine the maximum mass of urea that could be formed from 50.0 cm3 of 0.100 mol dm−3 potassium cyanate solution.
Urea can also be made by the direct combination of ammonia and carbon dioxide gases.
2NH3(g) + CO2(g) (H2N)2CO(g) + H2O(g) ΔH < 0
Predict, with a reason, the effect on the equilibrium constant, Kc, when the temperature is increased.
Suggest one reason why urea is a solid and ammonia a gas at room temperature.
Sketch two different hydrogen bonding interactions between ammonia and water.
The combustion of urea produces water, carbon dioxide and nitrogen.
Formulate a balanced equation for the reaction.
The mass spectrum of urea is shown below.
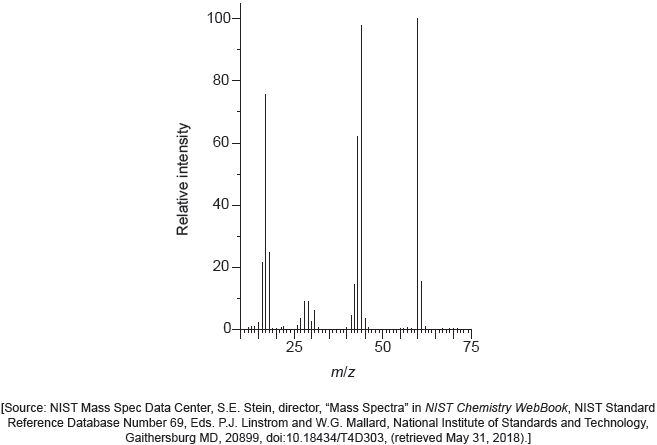
Identify the species responsible for the peaks at m/z = 60 and 44.
The IR spectrum of urea is shown below.
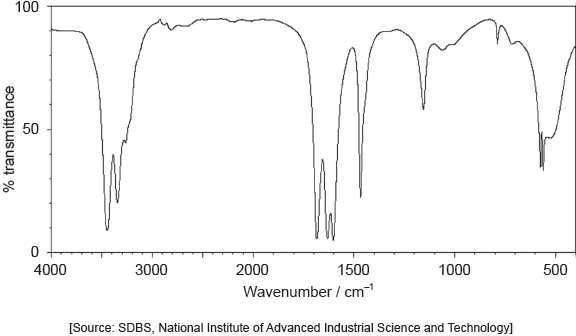
Identify the bonds causing the absorptions at 3450 cm−1 and 1700 cm−1 using section 26 of the data booklet.
Predict the number of signals in the 1H NMR spectrum of urea.
Markscheme
molar mass of urea «= 4 × 1.01 + 2 × 14.01 + 12.01 + 16.00» = 60.07 «g mol–1»
«% nitrogen = × 100 =» 46.65 «%»
Award [2] for correct final answer.
Award [1 max] for final answer not to two decimal places.
[2 marks]
«cost» increases AND lower N% «means higher cost of transportation per unit of nitrogen»
OR
«cost» increases AND inefficient/too much/about half mass not nitrogen
Accept other reasonable explanations.
Do not accept answers referring to safety/explosions.
[1 mark]
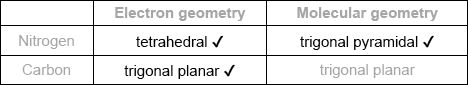
Note: Urea’s structure is more complex than that predicted from VSEPR theory.
[3 marks]
n(KNCO) «= 0.0500 dm3 × 0.100 mol dm–3» = 5.00 × 10–3 «mol»
«mass of urea = 5.00 × 10–3 mol × 60.07 g mol–1» = 0.300 «g»
Award [2] for correct final answer.
[2 marks]
«Kc» decreases AND reaction is exothermic
OR
«Kc» decreases AND ΔH is negative
OR
«Kc» decreases AND reverse/endothermic reaction is favoured
[1 mark]
Any one of:
urea has greater molar mass
urea has greater electron density/greater London/dispersion
urea has more hydrogen bonding
urea is more polar/has greater dipole moment
Accept “urea has larger size/greater van der Waals forces”.
Do not accept “urea has greater intermolecular forces/IMF”.
[1 mark]
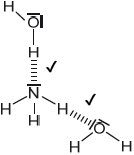
Award [1] for each correct interaction.
If lone pairs are shown on N or O, then the lone pair on N or one of the lone pairs on O MUST be involved in the H-bond.
Penalize solid line to represent H-bonding only once.
[2 marks]
2(H2N)2CO(s) + 3O2(g) → 4H2O(l) + 2CO2(g) + 2N2(g)
correct coefficients on LHS
correct coefficients on RHS
Accept (H2N)2CO(s) + O2(g) → 2H2O(l) + CO2(g) + N2(g).
Accept any correct ratio.
[2 marks]
60: CON2H4+
44: CONH2+
Accept “molecular ion”.
[2 marks]
3450 cm–1: N–H
1700 cm–1: C=O
Do not accept “O–H” for 3450 cm–1.
[2 marks]
1
[1 mark]
Examiners report
Compound A is in equilibrium with compound B.
Predict the electron domain and molecular geometries around the oxygen atom of molecule A using VSEPR.
The IR spectrum of one of the compounds is shown:
COBLENTZ SOCIETY. Collection © 2018 copyright by the U.S. Secretary of Commerce on behalf of the United States of America. All rights reserved.
Deduce, giving a reason, the compound producing this spectrum.
Compound A and B are isomers. Draw two other structural isomers with the formula .
The equilibrium constant, , for the conversion of A to B is in water at .
Deduce, giving a reason, which compound, A or B, is present in greater concentration when equilibrium is reached.
Markscheme
Electron domain geometry: tetrahedral ✔
Molecular geometry: bent/V-shaped ✔
B AND absorption/
OR
B AND absence of ✔
Accept any value between .
Accept any two isomers except for propanone and propen-2-ol:
✔✔
Penalize missing hydrogens in displayed structural formulas once only.
AND is greater than /large ✔
Examiners report
Half of the candidates answered correctly. The rest of the candidates often answered the question in terms of the carbon atom indicating that they did not read the question carefully.
About 50% of the candidates answered correctly. Quite a few, however, gave compounds other than A or B, indicating not reading the question properly or being confused by the skeletal formulas given in the question.
Nearly half of the candidates gave two correct isomers. Propanal was often given as one of the isomers. Some candidates repeated the compounds given in the question and a few gave structures with 5 bonds on a carbon atom.
Half of the candidates answered correctly. A common mistake was K > 0 instead of K > 1.
The properties of elements can be predicted from their position in the periodic table.
Explain why Si has a smaller atomic radius than Al.
Explain the decrease in radius from Na to Na+.
State the condensed electron configurations for Cr and Cr3+.
Describe metallic bonding and how it contributes to electrical conductivity.
Deduce the Lewis (electron dot) structure and molecular geometry of sulfur dichloride, SCl2.
Suggest, giving reasons, the relative volatilities of SCl2 and H2O.
Consider the following equilibrium reaction:
2SO2 (g) + O2 (g) 2SO3 (g)
State and explain how the equilibrium would be affected by increasing the volume of the reaction container at a constant temperature.
Markscheme
nuclear charge/number of protons/Z/Zeff increases «causing a stronger pull on the outer electrons» ✓
same number of shells/«outer» energy level/shielding ✓
Na+ has one less energy level/shell
OR
Na+ has 2 energy levels/shells AND Na has 3 ✓
less shielding «in Na+ so valence electrons attracted more strongly to nucleus»
OR
effective nuclear charge/Zeff greater «in Na+ so valence electrons attracted more strongly to nucleus» ✓
Accept “more protons than electrons «in Na+»” OR “less electron-electron repulsion «in Na+»” for M2.
Cr:
[Ar] 4s13d5 ✓
Cr3+:
[Ar] 3d3 ✓
Accept “[Ar] 3d54s1”.
Accept “[Ar] 3d34s0”.
Award [1 max] for two correct full electron configurations “1s22s22p63s23p64s13d5 AND 1s22s22p63s23p63d3”.
Award [1 max] for 4s13d5 AND 3d3.
electrostatic attraction ✓
between «a lattice of» cations/positive «metal» ions AND «a sea of» delocalized electrons ✓
mobile electrons responsible for conductivity
OR
electrons move when a voltage/potential difference/electric field is applied ✓
Do not accept “nuclei” for “cations/positive ions” in M2.
Accept “mobile/free” for “delocalized” electrons in M2.
Accept “electrons move when connected to a cell/battery/power supply” OR “electrons move when connected in a circuit” for M3.
H2O forms hydrogen bonding «while SCl2 does not» ✓
SCl2 «much» stronger London/dispersion/«instantaneous» induced dipole-induced dipole forces ✓
Alternative 1:
H2O less volatile AND hydrogen bonding stronger «than dipole–dipole and dispersion forces» ✓
Alternative 2:
SCl2 less volatile AND effect of dispersion forces «could be» greater than hydrogen bonding ✓\
Ignore reference to Van der Waals.
Accept “SCl2 has «much» larger molar mass/electron density” for M2.
pressure decrease «due to larger volume» ✓
reactant side has more moles/molecules «of gas» ✓
reaction shifts left/towards reactants ✓
Award M3 only if M1 OR M2 is awarded.
Examiners report
Hydrogen peroxide can react with methane and oxygen to form methanol. This reaction can occur below 50°C if a gold nanoparticle catalyst is used.
Methanol is usually manufactured from methane in a two-stage process.
CH4 (g) + H2O (g) CO (g) + 3H2 (g)
CO (g) + 2H2 (g) CH3OH (l)
Consider the first stage of the reaction.
CH4 (g) + H2O (g) CO (g) + 3H2 (g)
The diagram shows the Maxwell-Boltzmann curve for the uncatalyzed reaction.
Draw a distribution curve at a lower temperature (T2) and show on the diagram how the addition of a catalyst enables the reaction to take place more rapidly than at T1.
The hydrogen peroxide could cause further oxidation of the methanol. Suggest a possible oxidation product.
Determine the overall equation for the production of methanol.
8.00 g of methane is completely converted to methanol. Calculate, to three significant figures, the final volume of hydrogen at STP, in dm3. Use sections 2 and 6 of the data booklet.
Determine the enthalpy change, ΔH, in kJ. Use section 11 of the data booklet.
Bond enthalpy of CO = 1077 kJ mol−1.
State the expression for Kc for this stage of the reaction.
State and explain the effect of increasing temperature on the value of Kc.
Markscheme
curve higher AND to left of T1 ✔
new/catalysed Ea marked AND to the left of Ea of curve T1 ✔
Do not penalize curve missing a label, not passing exactly through the origin, or crossing x-axis after Ea.
Do not award M1 if curve drawn shows significantly more/less molecules/greater/smaller area under curve than curve 1.
Accept Ea drawn to T1 instead of curve drawn as long as to left of marked Ea.
methanoic acid/HCOOH/CHOOH
OR
methanal/HCHO ✔
Accept “carbon dioxide/CO2”.
CH4(g) + H2O(g) CH3OH(l) + H2(g) ✔
Accept arrow instead of equilibrium sign.
amount of methane = « = » 0.498 «mol» ✔
amount of hydrogen = amount of methane / 0.498 «mol» ✔
volume of hydrogen = «0.498 mol × 22.7 dm3 mol−1 = » 11.3 «dm3» ✔
Award [3] for final correct answer.
Award [2 max] for 11.4 «dm3 due to rounding of mass to 16/moles to 0.5. »
Σbonds broken = 4 × 414 «kJ» + 2 × 463 «kJ» / 2582 «kJ» ✔
Σbonds formed = 1077 «kJ» + 3 × 436 «kJ» / 2385 «kJ» ✔
ΔH «= Σbonds broken − Σbonds formed =( 2582 kJ − 2385 kJ)» = «+»197«kJ» ✔
Award [3] for final correct answer.
Award [2 Max] for final answer of −197 «kJ»
✔
Kc increases AND «forward» reaction endothermic ✔